Understanding Cross Sensitivities Can Help Keep Workers Safer
Toxic, combustible and asphyxiant gases pose some of the greatest threats to workplace safety. In some cases, you may be unsure which gases could be present in your work environment. Fortunately, many different options are available to detect gases in a facility. For example, you could collect a sample and have it analyzed with a mass spectrometer to get an exact reading of what is in the area. However, this method can take time, and if your onsite lab isn't properly equipped, you might have to send it out, which leads to additional expenses.
Alternatively, you can use colorimetric gas detector tubes, which are great because they only react with a specific chemical or a family of chemicals. The problem with tubes is that they can be expensive and some have a short shelf life. Additionally, if you are unsure of what the gas is, you could be cracking open multiple tubes until you find the appropriate one for the present chemical, so now you have wasted a lot of time and money.
Gas Detection Options
So what option do most people go for? The choice that usually makes the most sense is portable gas meters and/or area monitors. Equipped to detect a single gas or multiple gases, these monitors use sophisticated technologies to check for gas-related hazards. Catalytic diffusion and infrared (IR) sensors can detect combustible gases, while photo ionization detector (PID) sensors typically check for volatile organic compounds (VOCs) and other hydrocarbons. For toxic gases that can be harmful to the human respiratory system, the electrochemical sensor is the most practical option.
The basic components of an electrochemical sensor are a working (or sensing) electrode, a counter electrode, and usually a reference electrode. These electrodes are enclosed in the sensor housing in contact with a liquid electrolyte. The sensor has a Teflon membrane that allows gas to enter and helps prevent dirt, water and other contaminates from entering the sensor. The gas diffuses into the sensor and through the membrane to the working electrode.
When the gas reaches the electrode, a chemical reaction occurs, and during this reaction, energy is created. The newly created electric current is proportional to the gas concentration that reaches the sensor, i.e., more gas equals higher electrical current created, less gas equals lower current. The instrument then displays the gas concentration in parts per million (PPM) for toxic gas sensors and percent volume (% vol) for oxygen sensors. Each sensor is designed to be specific to the gas it is intended to detect, but we can get cross sensitivity with this sensor technology.
Understanding Cross Sensitivity
Cross sensitivities, also referred to as interfering gases, are gases that can cause the electrode inside the sensor to react even if the target gas is not present. Can this reaction be eliminated? In a perfect world, yes, but unfortunately, it is very difficult to develop an electrode that will not respond to anything other than the target gas.
Ideally, a user would want a carbon monoxide (CO) sensor to only read carbon monoxide, but there are other gases that can generate readings on this sensor. This is just one example of many cross interferences. We can slow this reaction down with the use of filters on the sensors, but yet again, it does not eliminate the reaction. At this time, there is not a perfect technology that can filter out all of the interfering gases.
So if you're the person in charge of a gas detection program, this can be problematic for you and your workers. They might be in an area where they are required to wear a CO single gas monitor and they get a gas reading on the meter. Now they know from their onboarding training when they got hired, that carbon monoxide is produced from incomplete combustion. They look around and they don't see anything that could be producing CO. You now have an uphill battle with the users since they no longer trust the meters because there is no CO in that area at all. But what really happened here? What the workers didn't realize is that the process area that they walked through had a large amount of hydrogen (H2) that caused the carbon monoxide sensor to respond. This is one of the reasons why training people up front about the potential for cross sensitivity is very important.
Investigating Gas Readings
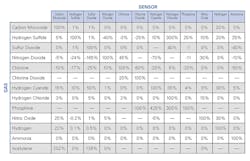
As the party that needs to investigate this gas reading, how can cross sensitivity help you? Being as important as it is, most gas detection manufacturers publish their toxic gas sensor cross sensitivities. In the sample chart shown (Figure 1), you can see that the sensor type is listed across the top, and the interfering gases are listed down the left-hand side. This chart could easily be printed as a reference for workers to keep in their pockets and take with them while working. If a worker has an instrument configured with a carbon monoxide, hydrogen sulfide, oxygen and an LEL sensor, he or she could get to work with this unit and the cross sensitivity chart. If the worker gets an unusual reading on the instrument like in the previous example, he or she may reference the chart to get a better understanding of the gases that could be present. Is this an exact science? No, not at all. But this method can be used in conjunction with another gas detection method that we talked about earlier.
If you or an end user gets positive readings of CO and H2S in an area where neither of these gases should be present, you can refer to the cross sensitivity chart to see which gases may have caused the readings. With the sample chart provided, you can now see it could be hydrogen cyanide (HCN) or maybe H2. Armed with this information, you are ready to grab your colorimetric tubes. Now, instead of grabbing a whole bunch and blindly cracking them open just to see what you might get, you can narrow it down to a family of chemicals or even a specific chemical.
Another thing to note with cross interference is that some monitors may show negative readings, depending on the manufacturer. This is another point of contention for end users. Sometimes they ask, "How do I get negative gas readings?" They then assume the meter must be broken. Negative readings occur when the gas that is present causes the electrode to react in a negative fashion. In this instance, it is cross sensitivity that causes the chemicals inside to react. Some manufacturers do what is called Dead Banding of the sensors in the sensor programming. This means that if the sensor is getting negative readings, it is programmed to not show this on the monitor's display. In this instance, you will just see a reading of zero. Some manufacturers do this in the low levels of the positive range as well to keep users from seeing low-level cross sensitivities and fluctuations. Other manufacturers choose not to dead band sensors, as the small fluctuations and negative readings can let a user know that something is in the area. As long as the meter is properly calibrated, you can assume that it is reading correctly.
Keeping Workers Safe
So, are cross sensitivities a bad thing? Sure, they can cause some headaches at times, but they can be helpful by letting you know that there is a gas present. Cross sensitivities can tell a user, "Hey, there is something here. You might want to look into it." And knowing what cross sensitivities apply to your units and your workplace can help you narrow down what gases could be present.
It is not a perfect science, but understanding cross sensitivities can certainly help to keep workers safe.
John Onuska III serves as a training specialist at Industrial Scientific, a provider of gas detection equipment.
About the Author
John Onuska
John Onuska is a training specialist with Industrial Scientific and a Lieutenant in the South Beaver Volunteer Fire Department in South Beaver, Pa. He can be reached at [email protected].